11. March 2026
The Liquid BBL: A Crisis Being Injected One Syringe at a Time
Why the non-surgical Brazilian Butt Lift is one of the most dangerous procedures available in the UK today — and why an immediate ban is long overdue
Written by Nurse Lee - Advanced Aesthetics Nurse Practitioner
I have worked in aesthetic medicine for a number of years. I have seen the industry evolve dramatically — from the early days of unregulated botulinum toxin being administered in beauty salons, through successive waves of new injectable products, to the hyper-commercialised, social-media-saturated landscape we find ourselves in today. Throughout all of that, I have never been as deeply alarmed by a single procedure as I am by the liquid Brazilian Butt Lift.
This post is not intended to be scaremongering. It is intended to be honest. And the honest truth is this: the liquid BBL — also known as the non-surgical BBL — is a procedure that is killing and seriously injuring people in the United Kingdom, being performed in completely unregulated environments by completely unqualified individuals, and being marketed to vulnerable women on social media as if it were no more risky than a facial treatment.
In February 2026, the UK Parliament's Women and Equalities Committee published a landmark report calling for an immediate ban on liquid BBL procedures. This is a position I support without reservation. In this post, I will explain exactly why — drawing on clinical evidence, patient safety data, and the regulatory failures that have allowed this crisis to escalate.
What Is a Liquid BBL — and Why Is It Considered 'Non-Surgical'?
A liquid BBL, or non-surgical Brazilian Butt Lift, is a cosmetic procedure designed to augment the size and shape of the buttocks without surgical fat transfer. It involves injecting large volumes of dermal filler — typically hyaluronic acid (HA)-based products, or biostimulators such as poly-L-lactic acid (PLLA) — directly into the buttock tissue to create the appearance of increased volume and projection.
The term 'non-surgical' is, in my professional opinion, deeply misleading. It implies a level of safety and simplicity that is entirely at odds with the clinical reality. The buttocks are a highly vascularised anatomical region containing major blood vessels — including the superior and inferior gluteal arteries — and any injection into this area carries the potential for catastrophic vascular injury (Cadogan Clinic, 2024).
Unlike facial filler treatments, where complications — whilst serious — can often be managed with prompt administration of hyaluronidase, complications from buttock filler frequently require hospital admission, intravenous antibiotics, surgical debridement, and intensive care intervention. In some cases, they are fatal.
A procedure involving the injection of up to 1,000 millilitres of filler into a richly vascularised anatomical region by someone with no medical training is not 'non-surgical'. It is a medical procedure being carried out in a clinical vacuum.
The Clinical Risks: What Can — and Does — Go Wrong
Vascular Occlusion and Tissue Necrosis
From a clinical standpoint, the single most serious immediate risk of any dermal filler injection is vascular occlusion — the inadvertent injection of filler into a blood vessel, or the compression of a vessel by surrounding filler material. When this occurs in the buttock region, the consequences can be far more catastrophic than in the face.
The Complications in Medical Aesthetics Collaborative (CMAC) — a group of UK-based clinicians who authored guidelines published in the Journal of Clinical and Aesthetic Dermatology — confirm that hyaluronic acid fillers can be introduced into any tissue plane, bringing with them the risk of inadvertent intravascular injection. If a vascular occlusion is not diagnosed and managed promptly, tissue necrosis will ensue (Murray et al., 2021). In the buttock region, the sheer volume of filler being injected — often hundreds of millilitres in a single session — dramatically elevates this risk compared with small-volume facial treatments.
Research published in the Journal of Cosmetic Dermatology by Wang et al. (2024) further confirms that vascular occlusion induced by HA injections can lead to severe adverse events, including necrosis, blindness, and cerebral infarction. Whilst the majority of documented cases in the literature concern the facial region, the underlying pathophysiology is identical when large volumes of filler are injected into vascular-rich tissue.
The key point — and this cannot be overstated — is that effective management of a vascular occlusion requires immediate recognition of ischaemic signs, prompt injection of high-dose hyaluronidase, and in many cases, surgical intervention. These are skills and resources that no unqualified practitioner operating outside a clinical setting will possess.
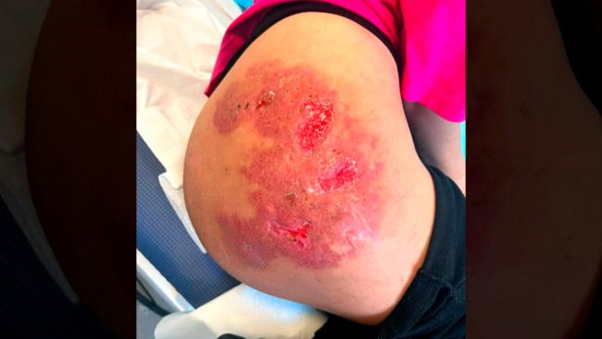
Sepsis and Life-Threatening Infection
The Save Face organisation — the government-approved register of qualified aesthetic practitioners — has been collecting and reporting data on liquid BBL complications since it launched its campaign to ban the procedure in December 2023. The figures are extraordinary.
Between December 2023 and January 2026, Save Face recorded 947 people who suffered severe complications following liquid BBL procedures in the UK. Approximately half of those affected were left fighting for their lives. Around 94% required hospital treatment, and 43% needed corrective surgery to manage the complications. The most common severe complications included sepsis, serious infections, and abscesses (Save Face, cited in The Fashion Central, 2026).
The case of Louise Moller, documented by Save Face, is illustrative. The 27-year-old travelled from Bolton to Essex to have the procedure performed by a non-healthcare practitioner. No consultation was conducted beforehand, and 500 millilitres of hyaluronic acid filler was injected into her buttocks in a single session. During the procedure, she experienced severe shooting pains. Her condition deteriorated rapidly, and she was taken to hospital by ambulance. Surgeons told her she had developed sepsis and required urgent surgical debridement to remove infected and necrotic tissue. Without that surgery, she was told she would have died within the hour. Weeks later, she remained in significant pain and required regular wound dressing changes by district nurses (Save Face, 2023).
This is not an isolated case. It is, tragically, representative of a pattern that has been replicated hundreds of times across the country.
Filler Migration and Long-Term Sequelae
Even in cases where immediate life-threatening complications do not occur, patients face significant long-term risks. The large volumes of filler used in liquid BBL procedures can migrate over time, causing asymmetry, hard nodules, chronic pain, and in some cases, the formation of granulomas — chronic inflammatory reactions to foreign material (Tayntons, 2025; Surgero, 2024).
Hyaluronic acid fillers in the gluteal region typically persist for 12 to 18 months, but migration and inflammatory responses can make correction extremely difficult and expensive. For non-reversible fillers — such as those based on calcium hydroxylapatite or PMMA — there is no dissolving agent available, meaning any complications must be managed surgically, potentially involving prolonged hospitalisation and multiple procedures.
Deep Vein Thrombosis and Pulmonary Embolism
The introduction of large volumes of foreign material into the lower limb region also raises the risk of deep vein thrombosis (DVT) and pulmonary embolism. These are well-recognised complications of surgical procedures in the gluteal and lower limb region, and there is no reason to believe that large-volume buttock injections are exempt from these haematological risks. In the absence of any pre-procedural assessment, thromboembolic risk stratification, or prophylactic measures, the risk to patients is entirely unmitigated (Tayntons, 2025).
Why 99% of Medical Professionals Refuse to Perform This Procedure
Save Face conducted research — published in December 2023 — revealing that 99% of medical professionals do not perform non-surgical BBL procedures because they consider them too dangerous. This is not timidity or excessive caution: it is an informed professional assessment made by clinicians who understand the anatomical risks involved, who are aware of their duty of care, and who recognise that the margin for error is essentially zero (Save Face, 2023).
The fact that a procedure is being widely performed — in Airbnbs, hotel rooms, garden sheds, and public toilets according to evidence heard by the Women and Equalities Committee — by individuals with no medical training, no understanding of vascular anatomy, no hyaluronidase to hand, and no ability to manage complications, is not merely a regulatory oversight. It is a public health emergency.
When 99% of qualified medical professionals decline to perform a procedure because they consider it too dangerous, the question we must ask is: who is performing it — and what does that tell us about the safety of the patient?
Unlike facial filler complications, where a skilled injector with the appropriate reversal agent can often intervene in time to prevent permanent damage, complications from buttock filler procedures frequently overwhelm the capacity of even well-equipped hospital teams. The sepsis cases documented by Save Face required intravenous antibiotics, surgical theatres, and ICU-level care. Not a hyaluronidase pen and a phone call to 111.

Alice Webb and the Human Cost of Regulatory Failure
Any discussion of the liquid BBL must acknowledge Alice Webb. On 24th September 2024, Alice — a 33-year-old mother of five from Gloucestershire — became the first person in the UK to die following a liquid BBL procedure. The procedure is understood to have been performed by a non-medic. Two individuals were subsequently arrested on suspicion of manslaughter (The Scotsman, 2024).
Alice Webb's death was described by Save Face as a tragedy that 'should never have been allowed to happen' — one that could and should have been prevented by the introduction of regulatory protections that had been called for years earlier (Save Face, 2024).
Tragically, Alice Webb was not the last person to die in connection with this procedure. In early 2026, Jordan Parke — a 34-year-old who had previously been arrested in connection with Alice Webb's death — reportedly died himself following a botched BBL procedure. These are not statistics. These are human lives, ending or being irrevocably damaged by a procedure that should never be taking place in unregulated settings (The Fashion Central, 2026).
It is also worth noting that at least one other British national had previously died as a result of a BBL procedure performed abroad, in Turkey, prior to Alice Webb's death. Cosmetic tourism adds an additional, and largely invisible, layer to the scale of this problem.
The Regulatory Vacuum: Over a Decade of Inaction
The regulatory situation in the UK is, to put it plainly, scandalous. In 2013, the then-head of the NHS issued a stark warning: a person undergoing a non-surgical cosmetic procedure had no more consumer protection than someone buying a toothbrush. That was over a decade ago. Since then, the only thing that has meaningfully changed is the scale of the harm being done (Women and Equalities Committee, 2026).
Currently, anyone in the UK — regardless of training, qualifications, or clinical experience — can legally perform a liquid BBL. There is no requirement for a clinical setting. There is no requirement for informed consent to be documented by a qualified professional. There is no requirement for the practitioner to carry reversal agents or emergency medication. There is no requirement for any post-procedure follow-up. This is the legal framework within which hundreds of patients have been hospitalised and at least one has died.
The Joint Council for Cosmetic Practitioners (JCCP) has been working for years to drive through a licensing framework for aesthetic practitioners, including a RAG (Red/Amber/Green) risk classification system that would restrict the highest-risk procedures to qualified medical professionals. Progress has been repeatedly stalled by ministerial delays, a General Election, and what the Women and Equalities Committee has described as a lack of urgency that is 'fostering complacency in self-regulation' within the industry (WEC, 2026).
In August 2025, the Department of Health and Social Care introduced measures intended to restrict the highest-risk non-surgical procedures — including liquid BBLs — to qualified healthcare professionals. This was a welcome, if long-overdue, step. However, the Women and Equalities Committee's February 2026 report concluded that this was insufficient and called for immediate, comprehensive action without further consultation (WEC, 2026).
The Women and Equalities Committee Report: A Watershed Moment
On 18th February 2026, the House of Commons Women and Equalities Committee published its eleventh report of Session 2024-26: Health Impacts of Breast Implants and Other Cosmetic Procedures (HC 869). It represents the most unequivocal parliamentary statement yet on the dangers of liquid BBLs and the failure of regulation to protect the public.
The Committee's key recommendations in relation to liquid BBLs are clear and urgent. 'High harm' procedures such as the liquid BBL should be banned immediately without further consultation. Liquid BBLs and liquid breast augmentations should be restricted to appropriately qualified medical professionals at once. Given the near-universal refusal of qualified medical professionals to perform these procedures, this restriction would constitute a de facto ban in all but the most exceptional clinical circumstances (WEC, 2026).
WEC Chair and Labour MP Sarah Owen stated in the report's foreword that individuals without any formal training are currently carrying out potentially very harmful interventions, often doing so in unsafe environments, and that 'this wild west of procedures is placing the public at risk' (WEC, 2026).
The report also called for a broader licensing framework for 'green' and 'amber' category lower-risk non-surgical cosmetic procedures to be introduced within the current Parliament. This is something the British Beauty Council, the Joint Council for Cosmetic Practitioners, Save Face, and numerous professional bodies have been calling for — it must be delivered without further delay (British Beauty Council, 2026).
'Regulation has not kept pace with the sector's expansion. In 2013, the head of the NHS warned that a person having a non-surgical cosmetic intervention has no more protection than someone buying a toothbrush. Over a decade later, the only thing that has changed is the number of people suffering life-changing and life-threatening injuries.' — Women and Equalities Committee, 2026
The Role of Advertising and Social Media
No analysis of the liquid BBL crisis is complete without addressing the role of social media advertising and the digital ecosystem that has normalised and glamourised this procedure. The Women and Equalities Committee's report explicitly links growing demand for cosmetic procedures to social media, influencer advertising, face-editing apps, and AI filters — all of which promote unrealistic body standards and incentivise vulnerable women to seek dramatic physical transformation (WEC, 2026).
In April 2025, the UK's Advertising Standards Authority (ASA) took action against six UK-based companies for advertising liquid BBL procedures irresponsibly. Using its AI-driven monitoring system to scan Facebook and Instagram, the ASA found that these advertisements exploited women's body image insecurities, used high-pressure time-limited tactics such as Black Friday deals, and made dangerously misleading claims — including a boasted '0% infection rate' and promises of 'safe, proven, and beautifully natural results' (ASA, 2025; Aesthetic Medicine, 2025).
Three of the six companies — did not even respond to the ASA's enquiries, displaying what the regulator described as an 'apparent disregard' for the advertising code. The ASA's spokesperson stated that choosing to undergo a cosmetic procedure is a serious decision, and that 'ads that trivialise this, exploit insecurities, or pressure consumers can cause real harm' (ASA, cited in The Guardian/AOL, 2025).
Ashton Collins, Director of Save Face, welcomed the ASA action but noted its limitations in the absence of formal regulation: these procedures are marketed as risk-free, painless, and inexpensive alternatives to surgery, but these claims are dangerously misleading. Save Face had by that point supported over 750 women who underwent liquid BBLs, with more than 55% having suffered from sepsis and more than 40% having required corrective surgery (Collins, cited in ASA/Guardian, 2025).
The advertising landscape continues to evolve faster than the regulatory one. For every advertisement the ASA bans, dozens more appear. Social media platforms bear a significant responsibility that they have, to date, largely declined to accept.
My Position as an Advanced Aesthetics Nurse Practitioner
I want to be clear about where I stand. I do not perform liquid BBL procedures. I will not perform liquid BBL procedures. And I will counsel every person who raises the subject with me — whether out of curiosity or serious intention — with the same clinical evidence I have laid out in this post.
In ethical aesthetic practice, the cornerstone of every decision is the benefit-risk assessment. We are obliged, under our professional and ethical duties, to only undertake procedures where there is a reasonable expectation that the benefit to the patient outweighs the risks. The liquid BBL, as currently performed in the UK — largely by unqualified individuals in unregulated environments, using large volumes of filler injected into a richly vascularised region with no capacity to manage complications — fails that test comprehensively.
I recognise that there are some practitioners who argue that in the hands of a highly skilled, medically qualified clinician, operating in an appropriate setting with full emergency protocols, limited-volume buttock filler has a role. That debate can continue in a regulated, evidence-based environment. But that is not what is happening on the ground. What is happening on the ground is that vulnerable women — many of them young, many of them influenced by an unforgiving social media culture that equates specific body shapes with worth and desirability — are being injected with industrial volumes of filler by people, in settings with no clinical oversight, and in some cases, they are dying.
That is not a grey area. That is a public health catastrophe, and it demands an urgent response.
Conclusion: The Government Must Act Now
The evidence against the liquid BBL is overwhelming, growing, and deeply distressing. We have 947 documented severe complications, hundreds of hospitalisations, multiple deaths, parliamentary condemnation, advertising bans, professional body campaigns, and a government watchdog calling for an immediate ban. What we do not yet have is the ban itself.
The Women and Equalities Committee has been explicit: there is no need for further consultation and delay. The Government must implement the WEC's recommendations without hesitation. Liquid BBLs must be banned immediately. A comprehensive licensing framework for all non-surgical cosmetic procedures must be established within this Parliament. And the broader cultural and digital forces driving vulnerable people towards high-risk procedures must be confronted — through media literacy education, responsible advertising standards, and honest public health communication.
As a clinician working in this field, I feel a professional and moral obligation to speak plainly and publicly about these risks. Patients who walk through the door of a reputable aesthetics clinic deserve to understand that there is a world of difference between evidence-based, regulated aesthetic practice and the unregulated, commercial free-for-all that constitutes much of the liquid BBL market. They deserve to know the truth — and the truth is that this procedure, as it stands, should not be available.
If you or someone you know is considering a liquid BBL, I urge you: please speak with a qualified, registered medical professional first. Do not rely on social media for clinical information. And please know that the 'non-surgical' label is not a synonym for 'safe'.
I also must clarify that I am no way involved or affiliated with any of the mentioned organisations within this article, they have purely formed part of my research and provide the relevant evidence for supporting claims against the procedure and my blog.
References
Advertising Standards Authority (ASA) (2025) ASA bans six liquid BBL advertisements for trivialising risks and exploiting body image insecurities. London: ASA. Available at: https://www.asa.org.uk (Accessed: 10 March 2026).
Aesthetic Medicine (2025) UK bans liquid Brazilian butt lift ads for risk trivialisation and body image pressure. Available at: https://aestheticmed.co.uk/uk-bans-liquid-brazilian-butt-lift-ads-for-misleading-claims (Accessed: 10 March 2026).
British Beauty Council (2026) Women and Equalities Committee Cosmetic Procedures Report. Available at: https://britishbeautycouncil.com/british-beauty-council-recommendations-are-included-in-women-and-equalities-cosmetic-procedures-report/ (Accessed: 10 March 2026).
Cadogan Clinic (2024) How to get a safe BBL: What is a liquid BBL? Available at: https://www.cadoganclinic.com/ask-the-expert/body-and-breast/fat-transfer-bbl (Accessed: 10 March 2026).
Murray, G., Convery, C., Walker, L. and Davies, E. (2021) 'Guideline for the management of hyaluronic acid filler-induced vascular occlusion', Journal of Clinical and Aesthetic Dermatology, 14(5), pp. E61-E69. Available at: https://pmc.ncbi.nlm.nih.gov/articles/PMC8211329/ (Accessed: 10 March 2026).
Save Face (2023) Urgent ban needed on dangerous liquid BBLs and breast augmentation procedures. Available at: https://www.saveface.co.uk/en/blog/post/urgent-ban-needed-on-dangerous-liquid-bbl-s-and-breast-augmentation-procedures (Accessed: 10 March 2026).
Save Face (2024) Non-surgical BBL and breast augmentation complaints and complications report. Available at: https://www.saveface.co.uk/en/blog/post/non-surgical-bbl-breast-augmentation-complaints-and-complications-report (Accessed: 10 March 2026).
Surgero (2024) What is liquid BBL: Risks and reasons to avoid. Available at: https://www.surgero.co/blog/what-is-liquid-bbl-risks-and-reasons-to-avoid (Accessed: 10 March 2026).
Tayntons (2025) Understanding the hidden dangers of BBL Brazilian butt lift procedures: A comprehensive guide. Available at: https://www.tayntons.co.uk/risks-of-brazilian-butt-lift-bbl-what-to-know-and-how-to-stay-safe/ (Accessed: 10 March 2026).
The Fashion Central (2026) Nearly 500 people in Britain came close to dying after liquid Brazilian butt lift procedures over the past two years. Available at: https://thefashioncentral.co.uk/nearly-500-brits-almost-died/ (Accessed: 10 March 2026).
The Scotsman (2024) What is a liquid BBL? Non-surgical procedure explained as mum dies. Available at: https://www.scotsman.com/health/what-is-a-liquid-bbl-non-surgical-butt-lift-explained-procedure-leads-death-mum-of-five-4798970 (Accessed: 10 March 2026).
Wang, Y., Liu, F., Zhang, Y. et al. (2024) 'Hyaluronic acid filler-induced vascular occlusion: Three case reports and overview of prevention and treatment', Journal of Cosmetic Dermatology, 23(2). doi: 10.1111/jocd.16147.
Women and Equalities Committee (WEC) (2026) Health impacts of breast implants and other cosmetic procedures: Eleventh report of session 2024-26 (HC 869). London: House of Commons. Available at: https://committees.parliament.uk/publications/51713/documents/286532/default (Accessed: 10 March 2026).
Disclaimer: This blog post is intended for educational and informational purposes only and does not constitute individual clinical advice. The views expressed are those of the Nurse Lee as an Advanced Aesthetics Nurse Practitioner. If you are considering any cosmetic procedure, please consult a qualified and registered medical professional before proceeding.